
5-Hydroxycytosine
| |
| Names | |
|---|---|
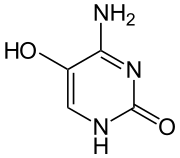
| Preferred IUPAC name
4-Amino-5-hydroxypyrimidin-2(1H)-one | |
| Other names
4-Amino-5-hydroxypyrimidine-2-one
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H5N3O2 | |
| Molar mass | 127.103 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
5-Hydroxycytosine is an oxidized form of cytosine that is associated with an increased frequency of C to T transition mutations, with some C to G transversions.[1] It does not distort the DNA molecule and is readily bypassed by replicative DNA polymerases.[2]

It has been shown in vitro to miscode for adenine.

5-hydroxycytosine is imperative for parallel DNA triplex formation, explaining why parallel triplexes form only at pH 6 and below.

References
- ^ Zahn KE; Averill A; Wallace SS; Doublié S (2011). "The miscoding potential of 5-hydroxycytosine arises due to template instability in the replicative polymerase active site". Biochemistry. 50 (47): 10350–10358. doi:10.1021/bi201219s. PMC 3280588. PMID 22026756.
- ^ Helmut Greim; Richard J. Albertini (2012). The Cellular Response to the Genotoxic Insult: The Question of Threshold for Genotoxic Carcinogens. Royal Society of Chemistry. p. 119. ISBN 9781849731775. Retrieved July 20, 2015.
See what we do next...
OR
By submitting your email or phone number, you're giving mschf permission to send you email and/or recurring marketing texts. Data rates may apply. Text stop to cancel, help for help.
Success: You're subscribed now !
